



When your whole world depends on productivity, every little bit counts
Adding Posilac® to your dairy management program allows healthy cows supplemented with Posilac® to produce an average of 10 more pounds of milk per day.1 This added productivity reduces your cost of production by [3%-5%, or approximately $0.50-$1.50/cwt.2 When you look at how milk production impacts your bottom line, Posilac® can make a world of difference.


Posilac® response begins with first supplementation and is maintained throughout lactation
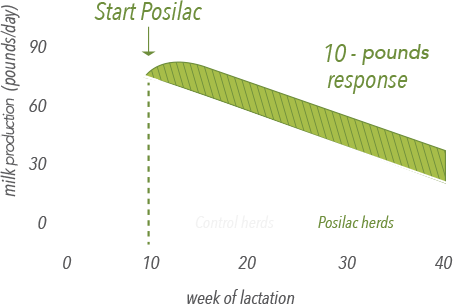
• A healthy cow supplemented with Posilac® produces an average of 10 more pounds of milk per day.1
• Response is consistent and sustained, lactation after lactation.1
• Herd production volume will continue to fluctuate, but the Posilac® response remains consistent.2
1. Ref. #1763. 10lb Executive Summary. UNION AGENER, INC., Data on File.
2. Bauman DE, et al. J. Dairy Sci. 1999;82(12):2564-2573

Increased productivity that impacts the future of your world.
For more than 20 years, Posilac® has enabled farmers to produce safe, nutritious milk that is affordable and is also produced in a [more environmentally responsible way].3
• All major global dairy importers allow the import and sale of US dairy products and meat from cows supplemented with Posilac®4
• Posilac® is the most researched animal product ever to be approved by the FDA4
• Regulatory authorities, together with their scientific assessment bodies in more than 50 countries, including Australia, Canada, the Commission of the European Communities (Committee for Veterinary Medicinal Products), South Korea, and the United States, have determined that milk and meat from cows supplemented with rbST are safe for consumption by people of all ages4.
A thriving dairy means everything to dairy producers and their families. What their farms produce is also vital to our future.
• The World Health Organization and FAO (Food and Agriculture Organization) have attested the product safety and his importance for the world's population6,7

rbST is a valuable management tool for use in dairy production to improve productive efficiency and to have less negative effects on the environment than conventional dairying. 5

Learn more about how you can put Posilac® to work in your herd.
Contact the Union Agener Customer Care Center
to place an order today.

Important Safety Information
The label contains complete use information, including cautions and warnings. Always read, understand, and follow the label and use directions.
Posilac®: For use in healthy lactating dairy cows. Supplement lactating dairy cows every 14 days beginning at 57–70 days in milk until the end of lactation.
Find more information about Posilac®
For a more customized report and to learn how Posilac® can lower your cost of production, e-mail us at dhall@unionagener.com
1 UNION AGENER, INC., Data on File
2 UNION AGENER, INC., Data on File
3 Capper J, Cady R, Bauman, D. The environmental impact of dairy production: 1944 compared with 2007. J Anim Sci. 2009;87:2160-2167.
4 Raymond R, Bales CW, Bauman DE, et al. Recombinant bovine somatotropin (rbST): a safety assessment. National Animal Interest Alliance Web site.
http://www.naiaonline.org/ pdfs/ Recombinant SomatotropinA Safety Assessment 2010.pdf Published March 22, 2010. Accessed May 12, 2014.
5 Judith L. Capper, Euridice Castañeda-Gutiérrez, Roger A. Cady, and Dale E. Bauman. The environmental impact of recombinant bovine somatotropin (rbST) use in dairy production.
6 FDA [Food and Drug Administration]. July, 2019. Bovine Somatotropin (bST). Available at https://www.fda.gov/animal-veterinary/ product-safety-information/bovine-somatotropin-bst. Accessed 17 December 2019
7 FAO [food agriculture organization] November, 2013 - Affirmation of the human food safety of recombinant bovine somatotropin by the Joint Food and Agriculture Organization of the United Nations (FAO)/World Health Organization (WHO) Expert Committee on Food Additives, Seventy-Eighth report, pages 70-78. Available at Link

Union Agener – Augusta, Geórgia
1788 Lovers Ln, Augusta
GA 30901 – United States of America
Privacy Policy |
Privacy Channel - LGPD (Brazil) |
Cookies Policy